 Toppers Study
Toppers StudyGet complete NCERT Solutions for 7 Science 6. Physical and Chemical Changes with detailed explanations. Easy-to-understand answers to help students study effectively and score better.

Get complete NCERT Solutions for 7 Science 6. Physical and Chemical Changes with detailed explanations. Easy-to-understand answers to help students study effectively and score better.
Preparing for exams becomes easier with Additional Questions With Solutions. Whether you are studying for board exams or mid-term exams, 7 Science Chapter 6. Physical and Chemical Changes solutions provide quick revising points, well-structured answers, and additional practice material to help you score better.
ncert_solutionsAdditional Questions With Solutions
Ques. – What do you understand by changes?
Ans. – When any object or material transforms its colour, size and taste etc that is known as changes. Ex – curd from milk, chapatti from Flour, chair from wood.
Ques. – What colour of light gives when ribbon of magnesium is burnt in air? What is called the Powderly ash obtained from this?
Ans. – It burns with a brilliant white light.
Ques. – What kinds are changes?
Ans. – There are two kinds of changes.
(1) Physical changes
(2) Chemical changes
Ques. – What are physical changes?
Ans. - A change in which a substance undergoes a change in its physical properties is called a physical change. Ex – Making any object from wood, Making kite of paper etc.
Ques. – What are chemical changes?
Ans. – A change in which one or more new substances are formed is called a chemical change. A chemical change is also called a chemical reaction. Ex- rusting, curd from milk.
Ques. – Name the changes in which no new substance is formed?
Ans. – physical changes.
Ques. - If you leave a piece of iron in the open for some time, it acquires a film of brownish substance.
(i) What is called this substance?
(ii) What is called this process?
Ans. –
(i) This substance is called rust.
(ii) The process is called rusting
Ques. – What new substance is formed when the ash of magnesium oxide is added in water?
Ans. – Magnesium hydroxide.
Ques. – Why does the blue colour of copper sulphate solution turn into green when an iron nail kept in?
Ans. – it occurs due to form a new substance iron sulphate.
Ques. – Write the chemical equation of reaction between carbon dioxide and lime water.
Ans. – The reaction between carbon dioxide and lime water is as follows:
Carbon dioxide (CO2 ) + Lime water [Ca(OH)2 ] ï‚®Calcium Carbonate (CaCO3 ) + Water (H2O)
Ques. – Write the complete equation for rusting?
Ans. – The complete equation for rusting is as follow;
Iron (Fe) + Oxygen (O2, from the air) + water (H2O)ï€ ï‚® ï€ rust (iron oxide Fe2O3)
Ques. – How would you show that setting of curd is a chemical change?
Ans. – Chemical change is the change in which there forms a new substance. Setting of curd is a new substance which is non reversible.
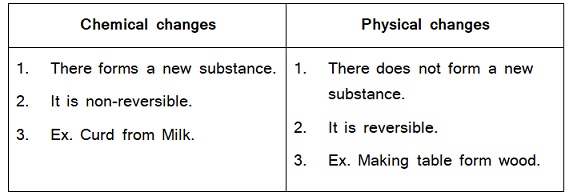
Ques. – Write the differences between chemical changes and physical changes.
Ans. –
Ques. – When baking soda is mixed with lemon juice, bubbles are formed with the evolution of a gas. What type of change is it? Explain.
Ans. – it is a chemical change. When lemon juice mixed with baking soda, acid of lemon juice reacts with baking soda (base) give salt and hydrogen gas, this cause forming bubbles.
Ques. - Explain why burning of wood and cutting it into small pieces are considered as two different types of changes.
Ans. – Burning of wood is chemical change in which forms new substances coal and carbon dioxide while cutting into small pieces is a physical change.
Ques. - Describe how crystals of copper sulphate are prepared?
Ans. – The crystals of copper sulphate are prepared by the process of crystallisation. In which there is added powder of copper sulphate in hot boiling water till highly concentrated. There obtains crystals after cooling the solution.
Additional Questions With Solutions are created by experts to give step-by-step explanations. Around 60–70% of exam questions are based on NCERT concepts. Our 7 Science Chapter 6. Physical and Chemical Changes solutions help you understand the core concepts and practice effectively.
Revision is the key to exam success. Our notes for 7 Science highlight important formulas, key definitions, and exam-ready points from Chapter 6. Physical and Chemical Changes. These quick revision notes make last-minute preparation easy.
Every NCERT chapter ends with exercises, and solving them is crucial. Our Additional Questions With Solutions include complete solutions for 7 Science Chapter 6. Physical and Chemical Changes exercises. With step-by-step answers, you gain clarity and confidence to attempt similar exam questions.
To boost your preparation, we also provide additional important questions with answers. These are prepared from previous year board papers, sample papers, and important concepts of Chapter 6. Physical and Chemical Changes. Practicing these ensures you are well-prepared for both board and mid-term exams.
Our Additional Questions With Solutions are useful for both board exams and mid-term exams. For 7 Science, we provide notes, exercises, and important Q&A so that you can revise smartly and write perfect answers in exams.
In short, Additional Questions With Solutions for 7 Science Chapter 6. Physical and Chemical Changes are a complete study package. With quick revising points, NCERT exercises, and additional important questions, you can prepare effectively for exams. Make these solutions your study companion and excel in your academic journey.
Go to other Class
Download worksheets and assignments for better practice and revision.